 
Universality of Physical Laws in Space and Time You can also see how the number of atoms in a given state Use the UNL Astronomy Education program's Hydrogenįurther explore how emission and absorption lines are created (link will appear Rainbow of colors of a continuous spectrum. High enough to where the smeared lines all merge together to produce the Of smearing, or broadening, depends on the density. Orange line is fattened so that one edge is in the yellow wavelengths and the This smears out the normally sharp spectral lines (they The energy levels of the atoms are distorted by their neighboringĪtom's electrons. Photons with energies between 1.89 and 3.4 eV not listed above will NOT be absorbed by the atom.Ī thermal spectrum is produced by atoms that are closely packed The line at 2.86 eV is for the n=2 to n=5 hop and the line at 3.02 eV is for the n=2 to n=6 hop. The spectrum shows the hydrogen absorption line energies. n=2 to unbound (ionizing the atom) if energy > 3.4 eV.n=2 to other energy levels higher than n=4 not shown in the figure found from formula: 13.6 x (1/4 - 1/n 2).n=2 to n=infinity at an energy of 0 – (-3.4) = 3.4 eV.The higher energy levels get too crowded together to distinguish them in the picture but here are the next two higher levels: n=5 at Energy E = -0.54 eV and n=6 at E = -0.38 eV. 
Only four of the many energy levels are shown in the figure below. Also, we will use the energy unit called “electron volt” (eV), where 1 eV = 1.602 x 10 -19 Joules to make the numbers easier to work with. In this convention, an electron with positive energy is free of the atom (unbound). (We'll see in chapter 11 that this is the case for A and B-type stars.) In this example, we will use the standard physics convention and have the values of the energy levels be negative numbers to mean that the electrons in the levels are bound to the atom. The absorption line depends on the density and temperature.įind the energies of the photons that can be absorbed by the hydrogen atom if the hydrogen gas is hot enough for its electron to usually be in the second energy level. Transition, the darker (or ``stronger'') the absorption line. (See the "How do you do that?" box below for a more detailed example of the absorption lines for hydrogen.) The more atoms The atom can absorb photons of just the rightĮnergy to move an electron from one energy level to another level. Photons of other energies pass right on by Less photons from the direction of the continuous source at that specific Necessarily in the same direction as the original photon! An observer will see The photon is later re-emitted but in a random 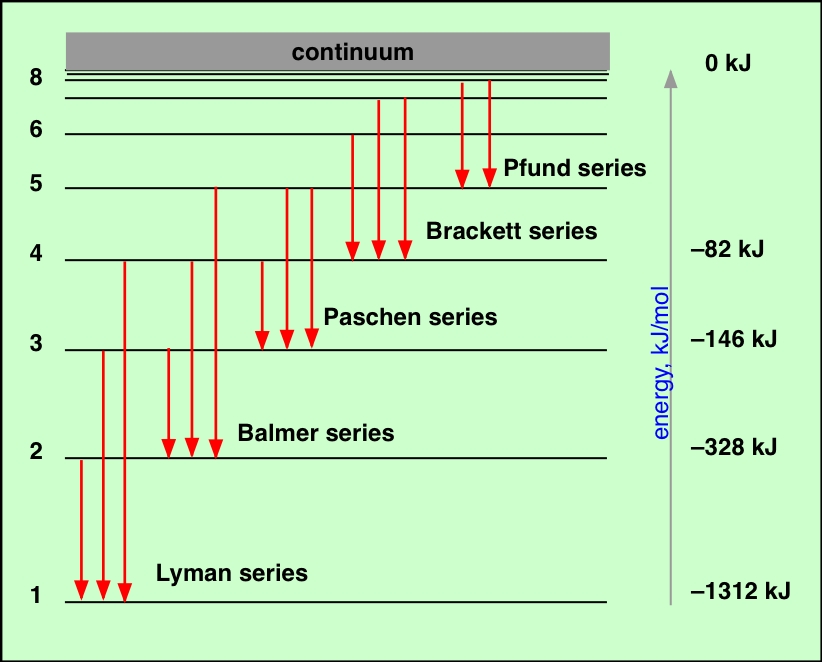
The photon is absorbed and electron moves toĮ 2. Sees a photon with energy E photon = E 2 -Į 1. They make up the rest of the continous spectrum you see.Įxample: An atom with electron in the E 1 orbit Photons moving through the gas with the wrong energy will pass right on by the atoms in Therefore, the pattern of absorption lines is the same as The size of the outward jumps made by the electrons are the same as the Photon had energy = the difference in energy of the energy orbits.īecause the energy levels in an element's atoms are fixed, Is absorbed by an atom, kicking an electron to a higher energy orbit. The intensity depends on the density and temperature ofĪn absorption line is produced when a photon of just the right energy The more atoms undergoing a particular transition, the more intense theĮmission line will be. The atom produces light of certain wavelengths. With greater energy (smaller wavelength). A larger jump to a lower energy level, will produce a photon Different jumps produce photons of differentĮnergies. Reach the ground state in one jump or it may temporarily stop at one or moreĮnergy levels on the way, but it canNOT stop somewhere between The energy of photon = the difference in energy of the energy orbitsĮxample: An atom with an electron at the E 2 orbit and wants The atom releases the energy is the form of a photon with that particularĮnergy. Remember rule #3! In order to go to a lowerĮnergy orbit, the electron must lose energy of a certain specificĪmount. Let's see how Bohr's model of the atom explains the three types of spectra.Īn emission line is produced by an atom in a ``excited'' energyĮnergy orbit as possible. Video lecture for this chapter This material (including images) is copyrighted!. Electromagnetic Radiation How Atoms Produce the Spectra Chapter index in this window 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
